Organic ChemistryReactions of Organic Compounds |
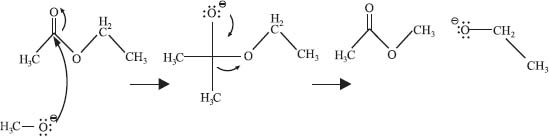
What are “curved arrows” in organic chemistry? |
Chemists used curved arrows to depict the flow of electrons in a chemical reaction. The arrow starts at the nucleophile (a lone pair of electrons, a π bond, or a σ bond), and points toward the electrophile (an atom, or bond, with a full or partial positive charge). Here’s an example of a transesterification reaction (i.e., changing one ester for another).