Sustainable “green” ChemistryIntroduction |
What was Agent Orange? |
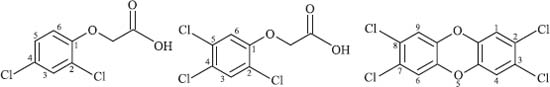
Agent Orange was an herbicide consisting of a mixture of 2,4-dichlorophenoxyacetic acid and 2,4,5-trichlorophenoxyacetic acid used by the U.S. military during the Vietnam War. The intent was to defoliate rural areas of the country, thus removing strategic ground cover and food sources from the rural areas. It was also discovered that the 2,4,5-trichlorophenoxyacetic acid was contaminated with 2,3,7,8-tetrachlorodibenzodioxin, which is an extremely toxic chemical. Agent Orange was sprayed throughout rural areas of southern Vietnam at high concentrations (an average concentration of thirteen times what was recommended by the USDA for domestic use), resulting in roughly 20% of southern Vietnam’s forests being sprayed. The use of the Agent Orange herbicide resulted in extremely negative health effects for people in these areas, and the effects still persist today despite the Vietnam War having ended in 1975. It is estimated that one million people are currently disabled or suffer major health problems as a result of the use of Agent Orange.
Chemical structures of 2,4-dichlorophenoxyacetic acid (left), 2,4,5-trichlorophenoxyacetic acid (middle), and 2,3,7,8-tetrachlorodibenzodioxin (right):